Today, February 17th, the US Food and Drug Administration (FDA) announced it is investigating consumer complaints of Cronobacter sakazakii and Salmonella Newport infections. All of the cases are reported to have consumed powdered infant formula produced from Abbott Nutrition’s Sturgis, Michigan facility.
As a result of the ongoing investigation, along with the US Centers for Disease Control and Prevention (CDC) and state and local partners, the FDA issued an alert to consumers to avoid purchasing or using certain powdered infant formula products produced at this facility.
This is an ongoing investigation, and the firm is working with the FDA to initiate a voluntary recall of the potentially affected product.
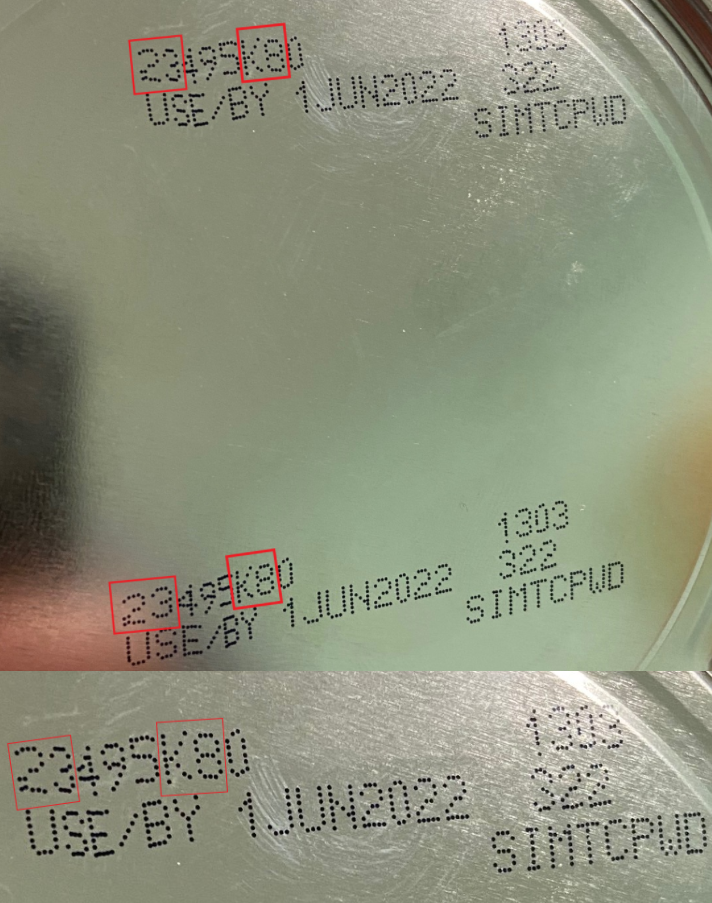
The FDA is advising consumers not to use Similac, Alimentum, or EleCare powdered infant formulas if:
- the first 2 digits of the code are 22 through 37; and
- the code on the container contains K8, SH or Z2; and
- the expiration date is 4-1-2022 (APR 2022) or later.
The FDA is investigating complaints of 4 infant illnesses from three states. All 4 cases related to these complaints were hospitalized and Cronobacter may have contributed to a death in 1 case. The FDA has initiated an onsite inspection at the facility. Findings to date include several positive Cronobacter sakazakii results from environmental samples taken by the FDA and adverse inspectional observations by the FDA investigators. A review of the firm’s internal records also indicates environmental contamination with Cronobacter sakazakii and the firm’s destruction of product due to the presence of Cronobacter.
Additional Information:
- Products made at the Sturgis, Michigan facility can be found across the US and were likely exported to other countries.
- Products that do not contain the information listed above are not impacted. The FDA advisory does not include liquid formula products or any metabolic deficiency nutrition formulas. Consumers should continue to use all products not covered by the advisory.
- To date, this investigation has been associated with four illnesses (three for Cronobacter and one for Salmonella) spanning the following states: Minnesota, Ohio and Texas. All 4 cases related to these complaints were hospitalized and Cronobacter may have contributed to a death in 1 case.
- Cronobacter bacteria can cause severe, life-threatening infections (sepsis) or meningitis (an inflammation of the membranes that protect the brain and spine). Symptoms of sepsis and meningitis may include poor feeding, irritability, temperature changes, jaundice (yellow skin and whites of the eyes), grunting breaths and abnormal movements. Cronobacter infection may also cause bowel damage and may spread through the blood to other parts of the body.
- Parents and caregivers of infants who have used these products, and are concerned about the health of their child, should contact their child’s health care provider. If your child is experiencing any of these symptoms, you should notify your child’s healthcare provider and seek medical care for your child immediately.
- Salmonella are a group of bacteria that can cause gastrointestinal illness and fever called salmonellosis. Most people with salmonellosis develop diarrhea, fever and abdominal cramps. More severe cases of salmonellosis may include a high fever, aches, headaches, lethargy, a rash, blood in the urine or stool, and in some cases, may become fatal.
- Parents and caregivers should never dilute infant formula and should not make or feed homemade infant formula to infants.
- If your regular formula is not available, contact your child’s healthcare provider for recommendations on changing feeding practices.
The FDA is continuing to investigate, and additional consumer safety information will be provided when it becomes available.
Discover more from Whatcom News
Subscribe to get the latest posts sent to your email.


